1īiallelic defects in the ADA gene cause deficiency of ADA, the enzyme that catalyzes the conversion of adenosine to inosine. However, measures are in place to monitor such events once the medicine is in use by using a registry of patients to study their long-term progress.In this issue of Blood, Reinhardt et al report the long-term clinical benefit and safety in patients with adenosine deaminase deficient severe combined immunodeficiency (ADA SCID) after gammaretroviral gene therapy using autologous bone marrow-derived CD34 + stem and progenitor cells. There is also a potential risk of autoimmune disease. Because Strimvelis is produced using a retrovirus, there could be a potential risk of cancer caused by unintended changes in the genetic material, although no such cases have been seen so far. 
Regarding safety, Strimvelis was relatively well tolerated although data are limited due to the small number of patients studied. Results from the main study show that Strimvelis is effective at improving survival of ADA-SCID patients. Strimvelis provides the opportunity of a cure that improves the working of the immune system for patients with ADA-SCID, which is a life-threatening condition. The Agency’s Committee for Medicinal Products for Human Use ( CHMP) decided that Strimvelis’s benefits are greater than its risks and recommended that it be approved for use in the EU. It must not be used in patients who have tested positive for human immunodeficiency virus (HIV, the virus that causes AIDS) or some other infections, or in patients who have previously had gene therapy treatment.įor the full list of restrictions and side effects reported with Strimvelis, see the package leaflet. Strimvelis must not be used in patients who have leukaemia (cancer of white blood cells) or myelodysplasia (a type of bone marrow disorder) or have had these conditions in the past. Serious side effects with Strimvelis may include effects linked to autoimmunity (when the immune system attacks the body’s own cells) such as haemolytic anaemia (low red blood cell counts due to their too rapid breakdown), aplastic anaemia (low blood cell counts due to damaged bone marrow), hepatitis (liver inflammation), thrombocytopenia (low blood platelet count) and Guillain-Barré syndrome (damage to nerves that can result in pain, numbness, muscle weakness and difficulty walking). The most common side effect with Strimvelis (which may affect up to 1 in 10 people) is pyrexia (fever). This type of medicine works by delivering genes into the body.īecause the number of patients with ADA-SCID is low, the disease is considered ‘rare’, and Strimvelis was designated an ‘ orphan medicine’ (a medicine used in rare diseases) on 26 August 2005. Strimvelis is a type of advanced therapy medicine called a ‘gene therapy product’. 
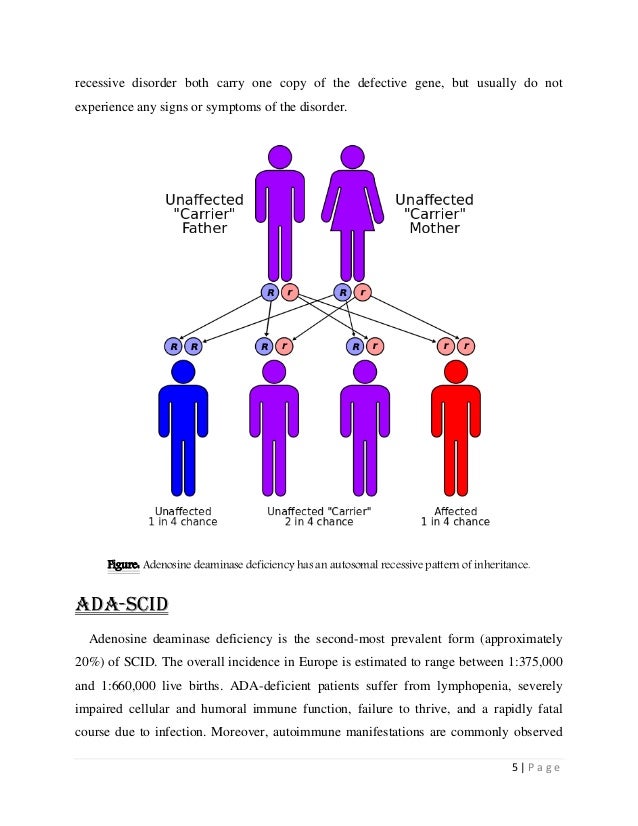
Some of the cells (called CD34+ cells) have been genetically modified to contain a working gene for ADA. Strimvelis contains cells derived from the patient’s own bone marrow. Strimvelis is used in patients with ADA-SCID who cannot be treated by a bone-marrow transplant because they do not have a suitable, matched, related donor. Because ADA is essential for maintaining healthy lymphocytes (white blood cells that fight off infections), the immune system of patients with ADA-SCID does not work properly and without effective treatment they rarely survive more than 2 years. As a result, patients lack the ADA enzyme. ADA-SCID is a rare inherited condition in which there is a change (mutation) in the gene needed to make an enzyme called adenosine deaminase (ADA). Strimvelis is a medicine used to treat severe combined immunodeficiency due to adenosine deaminase deficiency (ADA-SCID). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
